TECHNOLOGY
We will grow together with the best expertise and trust.
Hydrogen Water Story

Oxygen is the most important element of the body and life cannot live without oxygen (O2), according to the theory of Dr. Patrick Flanagan, an American genius scientist and medical scientist who discovered mega-negative hydrogen.
Hydrogen (H) is also the source of human life, and all living things are kept alive by hydrogen, and minus hydrogen (H−) has the key to life-death and aging.
This is the conclusion that life on Earth cannot be imagined without hydrogen ions.
Water (H2O) is the largest component of life.
A water molecule is a common combination of one Oxygen (O) atom and two Hydrogen (H) atoms, and oxygen atoms and hydrogen atoms share an electron pair, but the transcription pair is located on the side of an oxygen atom with a high electrical voice, the hydrogen atom has a negative charge and is highly polarized and the hydrogen atom has a strong concentration and is a strong object.
In order to drop the above coupled hydrogen molecules into the reactor, chemical and physical energy are used to break down the molecular structure of H2O combined with hydrogen and oxygen to break it down into OH and H or H2 and O, where the combination of OH is often divided into OH and H because it is stronger than H2.
Hydrogen molecules (H2) are known to be active hydrogen (H−) when they are released as hydrogen atoms (H) and they are elements close to our lives, and H2O combined with hydrogen and oxygen can be separated as much as possible.
If water is decomposed by an electric energy reaction, (+) plates can be oxygenated by oxidation reaction, and (-) reductively from the (-) plates to get hydrogen. The volume of hydrogen and oxygen is 2:1, and the overall reaction formula is as follows.
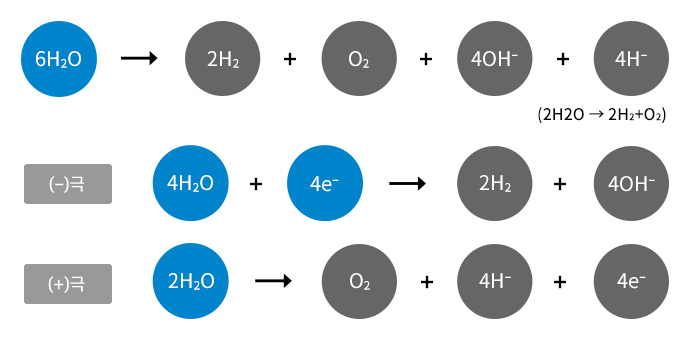
※ The hydrogen generator developed by the company is a method for converting electrical energy into chemical energy, which can easily be separated into hydrogen ions (H) and hydroxide ions (OHs) and allows separated ions to be combined with other ions by charge personnel.
What is hydrogen bonds?

Hydrogen molecules are not well separated from the reactor, but are separated for some reason and present in their respective atomic states, called activated hydrogen (H), and separated hydrogen is separated by positive and negative ion hydrogen.
Cationic hydrogen (H+) is present as a proton without electrons, while in the case of anionic hydrogen (H−) there are no protons and two electrons.
"H+"protones are highly reactive and dissipate momentarily (1/100 seconds), while "Hydrogen (H−)hydrite" is stable and combines with a mineral ion substance ionized in water to sustain energy for a long time.
In other words, hydrogen (H) is intended to be absorbed and combined in its mineral ion and win-win relationship. Therefore, in order to achieve hydrogen bonds, it is only absorbed in minerals when the reactor is separated, which is smaller and lighter than the hydrogen molecule (H2).
This is known as hydrogen bonds, and in order for hydrogen bonds to stabilize, hydrogen molecules must be nuclear reactors and at the same time be rich in ionized minerals in the water.
※ Measure the amount of hydrogen between 1000 and 2000 ppb using the hydrogen meter.If hydrogen bonds fail to form, hydrogen that disappears instantly becomes meaningless.
Hydroxide ion with reducing force
When water (H2O) is converted into electrical and chemical energy, it is decomposed into hydrogen (H) and hydroxide ions (OH−)
and separated ionic substances combine with other ionic substances in the water to produce various forms of matter.
Hydrogen ions (H−) separate from hydrogen are absorbed into the body by absorption into minerals, where hydrogen is released separately from the minerals.
Separated hydrogen ions (H−) turn the extra electrons over to the harmful oxygen (O2−) and turn them into oxygen (O2) needed for metabolic action or water to
return them to life-saving substances.
OHH (OH−) also strengthens anti-oxidant material production by turning high-oxidant water into a reduction power by electrically charged personnel.
When water (H2O) is decomposed, the concentration of an ion of hydroxide (OH 해리) is purged higher than that of hydrogen (H) ions, which makes it more alkaline and
thus adds one more negative electron to give it a strong reduction power with stable electronic arrangement.
In other words, OH ions make changes in the properties of water with high oxidizing power (Oxidation) to the potential of reducing power, while producing anti-oxidant →
Super-Oxide Disorder enzyme material for free radical removal.
In addition, antioxidants such as catarase, glutachion, puroxidase and peroxiretoxin will be raised in the body to benefit the body.
So, you're going to have to take the water out of electrolysis or chemo, and you're going to get it.
Hydrogen (H−) or hydroxide ions (OH−) produce anti-oxidant by the ORP action, reducing the free radical (Freeradical) oxide, which is beneficial to health life.
What is an oxidation potential?(ORP-ORP-Oxidation Redirection Potential)
An energy unit determined by the equilibrium between the oxidizer and the reducing agent present in the solution, the lower the electron reading is negative (-),
the stronger the return power, and the higher the electron reading is positive (+), the stronger the oxidation force.
Body fluids (water) in the body have very low levels of ORP.
This is due to the bio-principles of sustaining a negative (-100-250 mV) level of charge to the skin, including human organs, in order to stabilize the removal of
harmful substances from the body and oxidation (aging) inhibiting action, thus increasing the "natural healing antibody."
What is an antioxidant?
Bioactive enzyme substances that have antioxidant action, such as catarase, glutachion, puroxidase, and peroxiretoxin, are antioxidants that inhibit oxidation,
and are produced in the process of being converted into a reductive force.
This enables the generation of antioxidants (SOD...etc.) as water rich in hydrogen (H) generation is used.
Bioactivated enzyme (antioxidants) substances are very beneficial enzymes that quickly remove free radicals, which are harmful oxygen produced in the body.
What is an active oxygen?

Active oxygen is an active type of oxygen that is slightly different from oxygen in the chemical structure and is a byproduct of metabolism that is produced in the body.
Oxygen (O2) tends to maintain a chemically stable electronic distribution structure, usually with two electrons having a negative charge (-e).
However, if the electrons surrounding the atomic nucleus do not form a pair, lose a single electron, and remain a solitary electron, the oxygen atom becomes very aggressive and active,
attacking the atoms and atomic forces around it to regain their chemical stability.
Eventually, the oxygen taken from one electron is called an active oxygen, which causes a chain reaction to recover the lost electrons.
As naturally occurring by-products in the metabolism of oxygen (O2) that we breathe, superoxide ions, hydrogen peroxide, and free radicals act as active oxygen (hazardous substances).
Therefore, free oxygen is one of the necessary substances to protect our body. About 2 percent of the oxygen we breathe turns into active oxygen, protecting our body.
In order to break down pathogens and viruses that enter the body from the white blood cells, the immune cells release free radicals and kill harmful substances.
Therefore, highly reactive free radicals act as transformers to react and destroy cell tissues or bacteria.
Free radicals (Free Radical) react at random regardless of cell, causing excess free radicals to act as a terrible harm to the human body.
Causes of free radicals

In addition to being produced in the chemical reactions and energy metabolism of the human body, it occurs when the bloodstream recuperates, i.e. under stress and under intense exercise.
- When the blood is released from the internal organs after being stressed, the blood is re-treated, resulting in a large amount of free radicals that can damage the internal organs.
- In the case of exercise, blood is pumped through the heart and muscles, and after exercise, blood is pumped back into the digestive tract, creating free oxygen.
- Active oxygen generated by external factors is generated in large quantities by" self" radiation" air pollution" chemicals (cigarettes, pesticides, processed foods, chlorine compounds).
Effect of free radicals on the human body
The cells weak against oxidative stress caused by free radicals ① Endothelial cell and ② brain cells surrounding the inner wall of a vessel
Brain cells are being targeted at oxidative stress because they have a large oxygen demand that requires about a third of the oxygen we breathe.
Therefore, if the endocardial tissue surrounding the internal walls of the blood causes oxidation and various diseases, including aging, as an obstacle to the blood circulation.
This causative agent, the rapid removal of free radicals, shall be achieved.
For example, an active oxygen type, the ions (O2−), have additional negative (-) electrons in the oxygen, so they try to react with a positive (+) charge to stabilize themselves.
Since OH• is also to be combined against hydrogen (H) to convert it into water (H2O), it wants to react with materials with hydrogen (H) to stabilize it.
Water to eliminate the free radicals

Hydrogen reduced water, which is rich in negatively charged hydrogen (H⁻)
and hydroxide ions (OH⁻), is a chemical oxidation / reduction reaction in which
electrons are combined with active oxygen to exchange electrons.
Highly active oxygen is reduced to water (H₂O) and dissolved oxygen (DO).
This is due to the production of anionic hydrogen (H⁻) and hydroxide ions (OH⁻).
The ORP-Oxidation Reduction Potential action, which has a large negative
charge value, acts as an active oxygen by generating substances such as
bioactive enzymes (catalase, glutathione, peroxytase) in addition to the
SOD-Super oxide Dismutase enzyme. Remove it.
In other words, the anion hydrogen (H⁻) and hydroxide ions (H⁻)
substances having electrons in the excess are superoxide ions (O₂⁻)
hydrogen peroxide (H₂O₂), hydroxyl free radicals (OH •), etc. generated
by unsafe reduction of oxygen. In combination with the active oxygen
substances of the lasting biological stability.
※ The active hydrogen does not oxidize as opposed to the active oxygen, and the active hydrogen combines with the active oxygen to be reduced to water (H2O) and dissolved oxygen.
"H₂O→2H+O, H+OH+(H₂O₂)→H₂O"
The conclusion is a common theory among scholars that hydrogen reflux by minerals not only keeps health but also helps stabilize adult diseases because the free radicals (O2−),
which have extra electrons produced richly by hydrogen (H−) ions.
- The decontaminants of free radicals are hydrogen ions (H−) and hydroxide ions (OH−).
- High hydrogen ions (H−) produced by electrolysis benefit my body.
- Hydrogen recuperative water prevents aging and strengthens bio-recycling power by producing strong SODs and antioxidants.
※ Since our body is composed of 70% water, I am sure that not only 70% of dry mineral management but also 70% of damaged body recovery opportunities will be stabilized if water is selected well.
If hydrogen and reducing water continue to circulate inside the body, it will benefit the body by removing extra free radicals from the body without causing any side effects. KOREAN
KOREAN